Inoculation Mechanisms: Part One
Abstract
Grey cast iron with flak graphite precipitates carbon in the form of cementite and graphite during the process of solidification. The form of precipitated carbon has an effect on mechanical properties of grey cast iron out of which the most important are tensile strength and hardness.
Since pure Si and ferrosilicon are found to be ineffective as inoculants, their nucleation potency depends on the presence of minor elements such as Ca, Al, Zr, Ba, Sr, Ti etc. in the alloys.
In the production of quality cast irons the inoculation process is of vital importance. When comparing un-inoculated and inoculated irons, differences in microstructure are easily revealed, which again will strongly affect the final mechanical properties of the casting. Through inoculation, the graphite nucleation and eutectic undercooling of the iron can be controlled and this will be of crucial assistance in giving the iron its required service properties.
Grey cast iron with flak graphite precipitates carbon in the form of cementite and graphite during the process of solidification. The form of precipitated carbon has an effect on mechanical properties of grey cast iron out of which the most important are tensile strength and hardness. If cementite is present in perlite, we talk about perlitic grey cast iron. It is undesirable for cementite to be precipitated in the “free” form. The occurance of free precipitated cementite (This is cementite into ledeburite.) increases the hardness to a great extent and cast iron with this kind of microstructure is particularly difficult to machine.
There are many factors that influence precipitation of the ledeburitic cementite in the grey cast iron with flake graphite, such as input raw materials, chemical composition of the produced cast iron, furnaces for manufacturing the grey cast iron, the pouring temperature of casting, wall thickness, amount of inoculants,etc.The cooling speed is also essential and different speeds in cooling can in spite of the same chemical composition of the cast iron cause differently formed graphite and microstructure of the base.
If the cooling is carried out too fast or if the inoculation is carried out incorrectly, ledeburitic cementite can occur. The result is white solidification – solidification into ledeburite. Thin walled castings are very susceptible to precipitation of cementite from ledeburite. Solidification into ledeburite can be avoided if the grey cast iron is added in different modificators known as inoculants. A few examples of inoculents include the alloys of Fe-Si75Al 0.3 and CaSi, FeSiMn. Table 1 shows the chemical composition for some inoculant examples.
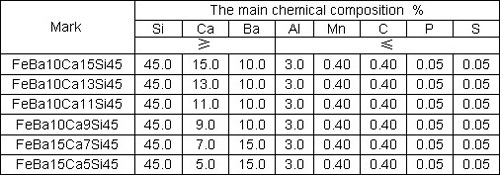
Table 1: The chemical composition of inoculant examples
Generally speaking complex inoculants which are in use today, besides silicon, also contain barium, zirconium, strontium and rare earths. The type of inoculant has a direct effect on formation of nucleation and dendrites of austenite.
Theories for Graphite Nucleation Mechanisms
As mentioned above, cast iron inoculants are based on ferrosilicon, graphite or calcium silicide, the former being the most common. Since pure Si and ferrosilicon are found to be ineffective as inoculants, their nucleation potency depends on the presence of minor elements such as Ca, Al, Zr, Ba, Sr, Ti etc. in the alloys. At present, the role of these minor elements are partly understood, but still complex matters related to formation of different types of nucleation sites in DI remain to be understood completely. Several theories that exist in the literature explain the phenomena of heterogeneous nucleation of graphite in solidifying cast iron. During the following passage, some of the most established theories are described and discussed.
The gas bubble theory
According to Karsay, graphite tends to crystallize onto any given surface or imperfections such as cracks, pinholes, inclusions, etc. The gas bubble theory states that graphite can form only if its crystallization is protected by the presence of some sort of phase boundary. The required phase boundaries are provided by the presence of carbon monoxide bubbles in the melt. The carbon monoxide bubbles are very finely dispersed in the melt and their size is less than 10μm. Karsay presented the gas bubble theory as illustrated in Figure 1.
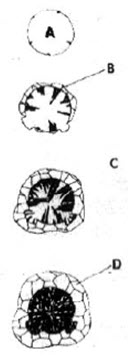
Figure 1: Karsay’s gas bubble theory is illustrated-(A) gas bubble, (B) graphite, (C) melt and (D) austenite.
The Graphite theory
The early theories for heterogeneous graphite nucleation are based on the assumption that the graphite nucleation occurred epitaxially from other graphite particles contained in the iron melt. Each extended these ideas to Si-based inoculants by proposing that their effectiveness is due to the formation of Si-rich regions around the dissolving particles within which the solubility of C is sufficiently reduced to promote graphite precipitation.
Later, Feest showed that this assumption is not correct, since the dissolution time of ferrosilicon in liquid iron is just a matter of seconds, and that graphite tends to form at the interface between the dissolving particle and the liquid. They therefore modified Eash’s model by proposing that these seed crystals will be preserved in the melt down to the eutectic temperature, provided that Sr or Ba is present in sufficient amounts to prevent redissolution of the graphite.
One weakness of the graphite theory and the assumption of small crystalline graphite particles, being preserved in the liquid iron for extended times, is the conflict with the well established fact that graphite in the form of crystalline recarburizers readily dissolves in liquid iron. Graphite recarburizers are typically added in sizes of millimeters, and will dissolve within seconds or a few minutes. Graphite based nucleation sites in a solidifying iron would be in the sizes of microns, and their dissolution time would consequently be very short. There is no question that graphite would be the ideal nucleation site for graphite itself. However, it can be argued whether the thermodynamic stability of micron sized graphite particles above the liquids temperature would withstand its own dissolution characteristics for the entire fading time of inoculation.
The Silicon Carbide Theory
Following the dissolution of ferrosilicon in liquid iron Wang and Fredriksson observed that silicon carbide crystals and graphite particles are formed in the melt close to the dissolving ferrosilicon particles. They also observed that these transient particles redissolve readily after the inoculation treatment. No oxide or sulphide particles are detected.
Based on their experimental observations, a theory developed and calculations were performed in order to explain the nucleation of graphite and the fading mechanisms. A salient assumption in Wang and Fredriksson’s model is the existence of an inhomogeneous distribution (local supersaturation) C and Si in the melt subsequent to the SiC dissolution which provides the necessary driving force for homogeneous nucleation of graphite.
The fading effect is thus explained by a homogenization of the melt with respect to Si and C through convection and diffusion. One weakness of the SiC theory for graphite nucleation is that the recognized critical role of elements like Ca, Sr and Ba in the FeSi inoculant cannot be explained by this theory. Another weakness of the SiC theory is the assumption of local supersaturation of C and Si due to restricted convection and diffusion.
Both C and Si are recognized for having very high diffusivity in liquid iron, and heat convection in hot metal is also recognized for being quite significant. It is therefore unlikely that dissolving SiC particles in Liquid iron would be capable of maintaining a supersaturation of C and Si throughout liquid metal processing and into the solidification. Furthermore, the observation of SiC and graphite, surrounding a partly dissolved FeSi inoculant particle, is most likely resulting from the experimental quenching technique itself, forcing transient SiC and C out of solution in the Si-rich metal during quenching.

Figure 2: Inoculants for iron.
Access Precise Properties of Cast Irons Now!
Total Materia Horizon contains property information for 11,000+ cast irons: composition, mechanical and physical properties, nonlinear properties and much more.
Get a FREE test account at Total Materia Horizon and join a community of over 500,000 users from more than 120 countries.