Clean Steel: Part Two
Abstract
Non-metallic inclusions, which are undesirable components of all steels, play an important role with respect to their effect on the steel properties. Controlling inclusions in steel is closely connected with the concept of "clean steel".
The improvement in steel properties by control of non-metallic inclusions plays an important part in defending the applications of steel against newer competitive materials.
Non-metallic inclusions, which are undesirable components of all steels, play an important role with respect to their effect on the steel properties. Controlling inclusions in steel is closely connected with the concept of “clean steel”. The improvement in steel properties by control of non-metallic inclusions plays an important part in defending the applications of steel against newer competitive materials. The aims of the metallurgist are to eliminate undesirable inclusions and control the nature and distribution of the remainder to optimize the properties of the final product.
Generally, non-metallic inclusions in steel normally have a negative contribution to the mechanical properties of steel, since they can initiate ductile and brittle facture. Among various types of nonmetallic inclusions, oxide and sulphide inclusions have been thought harmful for common steels.
All steels contain non-metallic inclusions to a greater or less extent. The type and appearance of these non-metallic inclusions depends on factors such as grade of steel, melting process, secondary metallurgy treatments and casting of steel. Because of this, it is of particular significance to determine how pure the steel is. The term steel cleanness is relative one, since even steel with only 1 ppm each of oxygen and sulfide will still contains 109 -1012 non-metallic inclusions per ton. From the viewpoint of “cleanness” all steels are “dirty”.
Non metallic inclusions in steel are the cause for dangerous and serious material defects such as brittleness and a vide variety of crack formations. However, some of these inclusions can also have a beneficial effect on steels properties by nucleating acicular ferrite during the austenite to ferrite phase transformation especially in low carbon steels. According to definition, the non-metallic inclusions are chemical compounds of metal with nonmetal which are present in steel and alloys like separated parts.
Classification of non-metallic inclusions
Non-metallic inclusions are divided by chemical and mineralogical content, by stableness/stability and origin. By chemical content non-metallic inclusions are divided into the following groups:
- Oxides (simple: FeO, MnO, Cr2O3, TiO2, SiO2, Al2O3 etc.; compound: FeOFe2O3, FeOAl2O3, MgOAl2O3, FeOCr2O3 etc.)
- Sulphides (FeS, MnS, CaS, MgS, Al2S3 etc.; compound: FeSFeO, MnSMnO etc.)
- Nitrides (simple: TiN, AlN, ZrN, CeN etc.; compound: Nb(C,N), V(C,N) etc, which can be found in alloyed steels and has strong nitride-generative elements in its content: titanium, aluminum, vanadium, cerium etc.)
- Phosphides (Fe3P, Fe2P etc.)
The majority of inclusions in steels are oxides and sulphides. Among various types of nonmetallic inclusions, oxide and sulphide inclusions have been thought harmful for common steels. Usually, nitrides are present in special steels (stainless steels, tool steels) which have elements with a strong affinity for nitrogen (e.g. chrome, vanadium), which create nitrides.
Figure 1 shows sulfides and oxides of non metallic inclusion in steel.

Figure 1: Non-metallic inclusion in steel: oxides-dark gray and sulfides-light gray
By mineralogical content oxygen inclusions are divided into the following groups:
- Free oxides – FeO, MnO, Cr2O3, SiO2 (quartz), Al2O3 (corundum) etc.
- Spinels-compound oxides which are formed by bi- and tri-valent elements as a ferrites, chromites and aluminates.
- Silicates which are presented in steel like a glass formed with pure SiO2 or SiO2 with admixture of iron, manganese, chromium, aluminum and tungsten oxides and also crystalline silicates.
Depending on the melting temperature, in liquid steel non-metallic inclusions are in solid or liquid condition.
As mentioned above the majority of inclusions in steels are oxides and sulfides. Sulfides in steel have been paid much attention because their treatment is an important problem in the steelmaking process. They affect on the properties of the final products by their deformation during the steel working process; especially their morphology has a significant effect on the steel properties.
According to analysis based on the steel ingots containing 0.01-0.15% S, the morphology of MnS can be classified into three types:
1) Type I is a globular .MnS with a wide range of sizes, and is often duplex with oxides.
2) Type II has a dendritic structure and is often called grain-boundary sulfide because it is distributed as chain-like formation or thin precipitates in primary ingot grain boundaries.
3) Type III is angular sulfide and always forms as monophase inclusion.
Most of the above mentioned sulfides are formed both during the process of secondary metallurgy or the solidification process. Recently, with the development of steelmaking technology, the sulfur concentration in steel was lowered drastically. Also, the continuous casting technology of steels with higher cooling rate than the ingot casting almost replaced the ingot casting.
So, the sulfides in the modern commercial steel are usually formed on solidification process or in solid steel during the subsequent cooling process. For example, the Widmanstätten plate-like MnS2, is formed in solid steel and Figure 2 shows the common morphology of MnS in conventional continuously casting steel, including the globular duplex oxide–sulfide (particle A, B and C) and the Widmanstätten plate-like MnS (particle D).

Figure 2: Typical duplex oxide–sulfide inclusion (particle A, B and C) and plate-like MnS (particle D) in conventional continuous casting silicon steel.
Numerous examples of the effect of non-metallic inclusions on steel properties show the importance of the behavior of the inclusions as well as of surrounding metal matrix during plastic working of steels. The aims of the metallurgist are to eliminate undesirable inclusions and control the nature and distribution of the remainder to optimize the properties of the final product.
An attempt by using program ABACUS was performed to model the behavior of slag inclusions and their surrounding matrix material during hot rolling and hot forging of hardenable steels. It is shown that it can be helpful for studying the behavior of inclusions, which is difficult or even impossible to obtain from a conventional experiment.
Figure 3 shows the effective strain contour during plastic deformation. Three regions of strain concentration (red) can be seen and a trihedral void (white region) close to the round inclusion is formed. The strain concentrations arise at the inner surface of the matrix. Another interested thing is that two edges of the pore tend to emerge and a bonding is formed. The difference in mechanical properties between the matrix and the inclusion is found to be the primary reason to create a void. The weak bonding at the interface between the matrix and the inclusion seems to facilitate to open the void.
Figure 4 shows the effect of rolling temperature on the relative plasticity index during hot rolling of steels. The relative plasticity index of inclusion increases while the rolling temperature rises. There exists a transition region, where the relative plasticity index changes rapidly. This trend agrees with the existing experimental results.
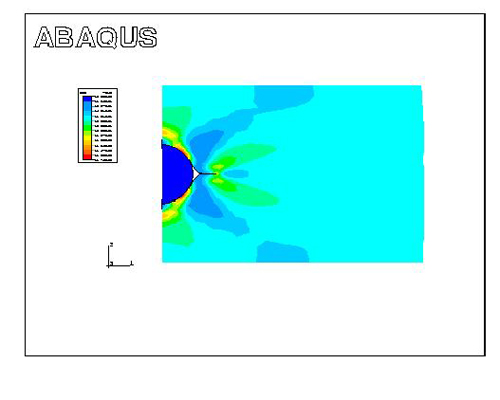
Figure 3: Void formation close to the inclusion.
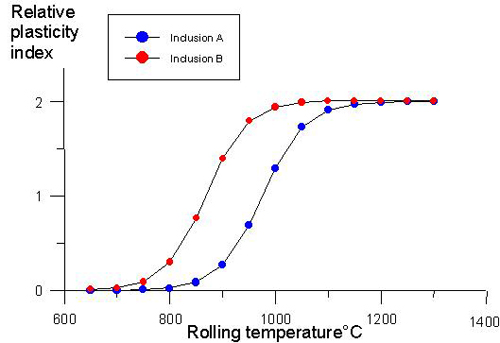
Figure 4: Effects of rolling temperature on the relative plasticity index.
Read more
Find Instantly Precise Compositions of Materials!
Total Materia Horizon contains chemical compositions of hundreds of thousands materials and substances, as well as their mechanical and physical properties and much more.
Get a FREE test account at Total Materia Horizon and join a community of over 500,000 users from more than 120 countries.